For example, this means that buying 1.5l bottle of mineral water, we need to bring home about 1.5 kg, etc. This approximation is very practical (and generally sufficiently accurate for everyday activities), because then 1l of water weighs approximately 1 kg. In everyday life, it is often assumed to approximate the water density to 1000 k g / m 3 kg/m^3 k g / m 3.Solve any question of Thermal Properties Of Matter with:. 
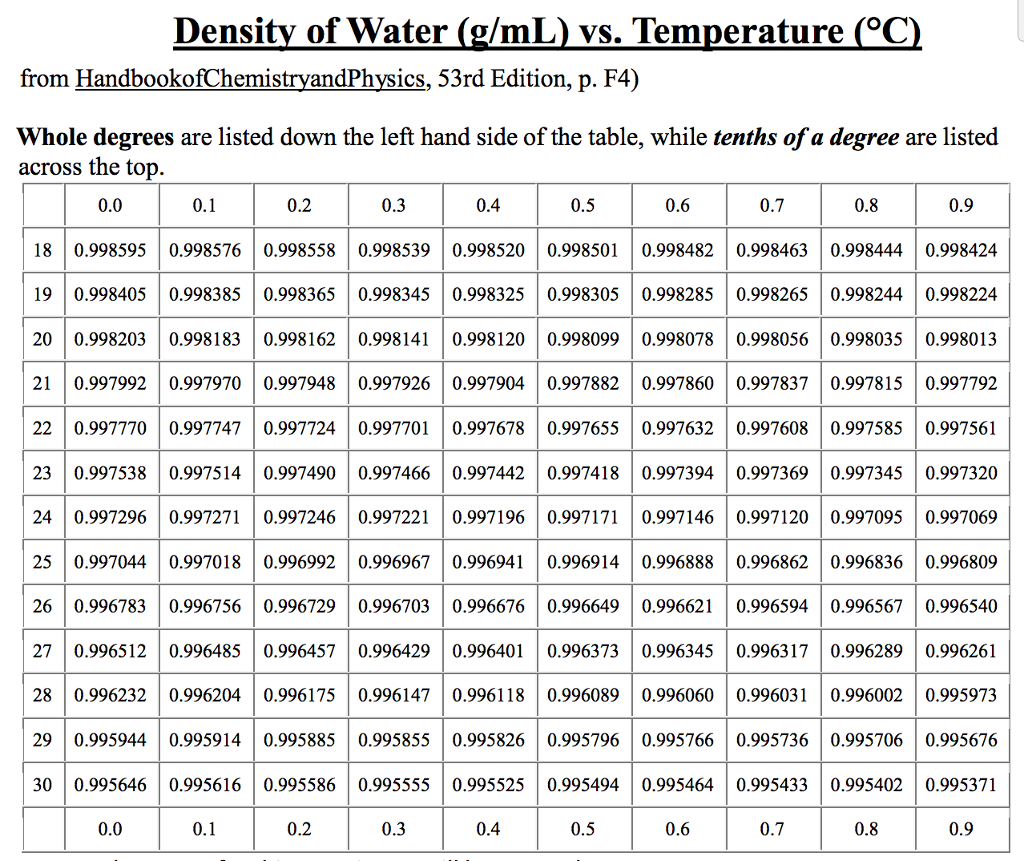
Water has the maximum density of 1g/cm3 only when it is pure water. When the temperature changes from either greater or less than 4 degrees, the density will become less than 1g/cm3. in the so-called normal conditions), the water density is equal to 1000 k g / m 3 kg/m^3 k g / m 3. Water has its maximum density of 1g/cm3 or 1000kg/m3 at 4C. Both below and above this temperature, the density of water is lower. The density of water reaches its maximum around 4☌.For some substances this effect is significant, in case of others it may be small. How the temperature influences density changes depends on the kind of substance. Above 4 ☌, however, thermal expansion becomes the dominant effect, and water near the boiling point (100 ☌) is about 4 less dense than water at 4 ☌ (39 ☏).The density of the substance varies depending on external conditions, i.e. Appendix II.Density and Viscosity of Water 0 C40 C Density, in Kinematic viscosity, Kinematic viscosity, Temperature, in Temperature, in Density, in grams pounds-mass per in square centimeters in square feet per degrees Celsius degrees Fahrenheit per cubic centimeter cubic foota per second second ×105 0 32 0.9998 62.42 0.0179 1.Other acceptable names are dihydrogen monooxide or oxydan. The molecular formula of water is H 2O and structural formula is H-O-H.At this temperature, a gallon of water weighs around 8.345 lbs. That is the reason why ice floats in water. However between 4 o and 0 o it becomes slightly less dense and when it freezes into ice the density becomes considerably less. Standard XII Physics Question The density of water at 4C is 1000kgm3 and at 104C it is 958.4kgm3. Water has a peculiar property that when it is cooled from temperatures above 4 o C it becomes more dense. The maximum density of water occurs at 4 ☌ because of the balance between the formation of clusters and the increase of empty space. Handbook Of Chemistry and Physics Online. Water has a maximum density of 39.2✯ or 4✬. Properties of Water Near 4 Degrees Celsius. Click the help icon for more information on the differences between Individual User accounts and Workspace accounts. This is equal to a rounded value of 1 gram per milliliter (g/ml) or 1 gram per cubic centimeter (g/cm 3) or 1000 kg/m 3. The water from chemical of view is a chemical compound (molecule) built of two hydrogen atoms and one oxygen atom. The density of water at 4 degrees Celsius is 8.345 lbs/U.S. The density of water is 1.940 sl/ft 3 at 39 F (4 C), and the specific weight in Imperial units is 1.940 sl/ft 3 32.174 ft/s 2 1.940 lb f /(ft/s 2 ft 3 ) 32.174 ft/s 2 62.If you heat it or cool it, it will expand. The pressures are stated in mega-Pascals, where a Pascal is a Newton per square meter, and as a multiple of standard atmospheric pressure. 4 degrees C turns out to be the temperature at which liquid water has the highest density. Statement 2: Relative density of a substance is the ratio of the density of the. Find out the exact value of water density at 4C (40F) and see a photo gallery of water properties. Water Vapor and Vapor Pressure Saturated Vapor Pressure, Density for Water Tempīelow are some selected values of temperature and the saturated vapor pressures required to place the boiling point at those temperatures. Which of the following statement (s) is/are true about relative density Statement 1: Relative density is known as the specific gravity of the substance. Learn how water density changes with temperature and ice is less dense than water.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |

 RSS Feed
RSS Feed
